Radiation Heat Transfer Examples and Applications
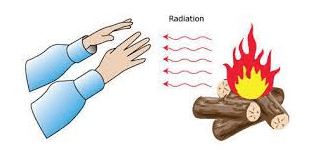
Radiation heat transfer is the mode of transfer of heat from one place to another in the form of waves called electromagnetic waves. Convection and conduction require the presence of matter as a medium to carry the heat from the hotter to the colder region. Some common examples of Radiation are Ultraviolet light from the sun, heat from a stove burner, visible light from a candle, and X-rays from an X-ray machine.
All life on Earth depends on the transfer of energy from the Sun, and this energy is transferred to the Earth over empty space. This form of energy transfer is the radiation of heat.
Our sun is the major source of heat energy. But how does this heat energy reach the Earth? It reaches us neither by conduction nor by convection because the space between the sun and the Earth’s atmosphere is empty. There is a third mode called radiation by which heat travels from one place to another. It is through radiation that heat reaches us from the sun.
See Also: Types of radiation
Radiation Examples
Here is a group of examples of Radiation in Everyday life:
- The transmission of electromagnetic waves through the microwave oven.
- The heat emitted by a radiator.
- Solar ultraviolet radiation is precisely the process that determines the Earth’s temperature.
- The light emitted by an incandescent lamp.
- The emission of gamma rays by a nucleus.
How does heat reach us directly from a fireplace?
Heat does not reach us by conduction through the air from a fireplace because air is a poor conductor of heat. Heat does not reach us by convection because the air getting heat from the fireplace does not move in all directions. Hot air moves upward from the fireplace. The heat from the fireplace reaches us directly by a different process in the form of waves called radiation. A sheet of paper or cardboard kept in the path of radiation stops these waves to reach us.
Radiations are emitted by all bodies. The rate at which radiations are emitted depends upon various factors such as:
- Colour and texture of the surface
- Surface temperature
- Surface area
Why does a cup of hot tea become cold after sometime?
All the objects, lying inside a room including the walls, roof, and floor of the room are radiating heat. However, they are also absorbing heat at the same time. When the temperature of an object is higher than its surroundings then it radiates more heat than it is absorbing. As a result, its temperature goes on decreasing until it becomes equal to its surroundings. At this stage, the body is giving out the amount of heat equal to the amount of heat it is absorbing.
When the temperature of an object is lower than its surroundings, then it is radiating less heat than it is absorbing. As a result, its temperature goes on increasing until it becomes equal to its surroundings. The rate at which various surfaces emit heat depends upon the nature of the surface. Various surfaces can be compared using Leslie’s cube.
Emission and Absorption of Radiation:
A Leslie cube is a metal box having faces of different natures.
The four faces of Leslie’s cube may be as follows:
- A shining silvered surface
- A dull black surface
- A white surface
- A colored surface
Hot water is filled in Leslie’s cube and is placed with one of its faces towards a radiation detector. It is found that a black dull surface is a good emitter of heat.
The rate at which various surfaces absorb heat also depends upon the nature of those surfaces. For example, take two surfaces, one is dull black and the other is a silver polished surface with a candle at the middle of the surface.
It is found that A dull black surface is a good absorber of heat as its temperature rises rapidly.
A polished surface is a poor absorber of heat as its temperature rises very slowly.
It is also found that the transfer of heat by radiation is also affected by the surface area of the body emitting or absorbing heat. The larger the area, the greater will be the transfer of heat. It is due to this reason that large numbers of slots are made in radiators to increase their surface area.
What is Thermal Radiation?
“Radiation given off by a body because of its temperature is called thermal radiation.”All bodies not only emit such radiation but also absorb it from their surroundings. If a body is hotter than its surroundings it emits more radiation than it absorbs and tends to cool. Normally, it will come to thermal equilibrium with its surroundings, a condition in which its rates of absorption and emission of radiation are equal. The spectrum of the thermal radiation from a hot solid body is continuous, its details depending strongly on the temperature.
If we were steadily to raise the temperature of such a body, we would notice two things:
- The higher the temperature, the more thermal radiation will be emitted. At first, the body appears dim, then it glows brightly: and
- The higher the temperature, the shorter the wavelength of that part of the spectrum radiating the most intensity. The predominant color of the hot body shifts from dull red through bright yellow-orange to bluish “white heat”.Since the characteristics of its spectrum depend on the temperature, we can estimate the temperature of a hot body, such as a glowing steel ingot or a star, from the radiation it emits. The eye sees chiefly the color corresponding to the most intense emission in the visible range.
The radiation emitted by a hot body depends not only on the temperature but also on the material of which the body is made, its shape, and the nature of its surface. For example, at 2000 k a polished flat tungsten surface emits radiation at a rate of 23.5 W/cm².
How can the temperature in a greenhouse be maintained?
Light from the sun contains thermal radiations (infrared) of long wavelengths, as well as light and ultraviolet radiations of short wavelengths. Glass and transparent polythene sheets, allow radiations of short wavelengths to pass through easily but not long wavelengths of thermal radiations. Thus, a greenhouse becomes a heat trap. Radiations from the sun pass easily through the glass and warm up the objects in a greenhouse.
These objects and plants give out radiations of much longer wavelengths. Glass and transparent polythene sheets do not allow them to escape easily and are reflected back in the greenhouse. This maintains the inside temperature of the greenhouse. The greenhouse effect promises better growth of some plants.
Carbon dioxide and water also behave in a similar way to radiations as glass or polythene. Earth’s atmosphere contains carbon dioxide and water vapors. It causes the greenhouse effect and thus maintains the temperature of the earth. In recent years, the percentage of carbon dioxide has increased considerably.
This has caused an increase in the average temperature of the Earth by trapping more heat due to the greenhouse effect. This phenomenon is known as Global warming. This has serious implications for global climate change.
Consequences of radiation
Different objects absorb different amounts of heat radiations falling upon them reflecting the remaining part. The amount of heat absorbed by a body depends upon the color and nature of its surface. A black and rough surface absorbs more heat than a White or polished surface.
Since good absorbers are also good radiators of heat. Thus, a black-colored body gets hot quickly absorbing heat reaching it during a sunny day and also cools down quickly by giving out its heat to its surroundings. The bottoms of cooking pots are made black to increase the absorption of heat from the fire.
Like light rays, heat radiations also obey the laws of reflection. The amount of heat reflected from an object depends upon its color and the nature of the surface. A white surface reflects more than colored or black surfaces. Similarly, polished surfaces are good reflectors than rough surfaces, and the reflection of heat radiations is greater than the polished surface.
Applications of Radiation of heat in everyday life
Every object emits or radiates some amount of heat. Knowledge of radiation can help us in many ways.
- When we sit beside a fire, the heat of fire reaches us by radiation.
- The cooling fins at the back of our refrigerator need to radiate their heat quickly to the surroundings. Its surface is made rough and painted black.
- During hot summer days, it is advised to wear white or light-colored clothes. White color absorbs less heat than dark colors.
- In cold areas, a greenhouse is used for the better growth of plants. Radiation from the sun passes through the glass or plastic and warms up the soil and plants. Plants and soil absorb and emit radiation and increase the temperature in the greenhouse. Plants grow well in the increased temperature of the greenhouse.
- Vacuum flasks are designed to keep liquids hot by minimizing heat losses in four possible ways conduction, convection, radiation, and evaporation. Conduction and convection through the sides of the flask are prevented by a vacuum between the double glass walls of the flask. Conduction through the trapped air above the liquid is minimum since air is a very poor conductor of heat. The stopper is usually made of plastic which is also a poor conductor of heat.
Evaporation and convection occur only when the plastic stopper is removed during use. Heat loss by radiation is harder to stop as radiant heat can pass through a vacuum. To minimize heat loss through radiation, the walls of the glass are silvered so as to reflect the radiant heat back into the hot liquid.
- Teapots, the shiny surfaces are bad emitters of radiation, shiny teapots can keep tea warmer than a black teapot for a longer time.
- The greenhouse is also working on the principle of radiation in which heat is trapped during winter to get warm inside the room.
Handpicked links
hi there buddy
hello..!
Hello!
hello. how may I help you?
hello